What is solubility in chemistry? It is a chemical property that helps to explain how and why certain substances mix, while others remain separate.
Table of Contents
1. Introduction
Let us make lemonade at home.
- Take a lemon and squeeze it into a glass to get lemon juice.
- Add some water and sugar to it.
- Stir it properly with a spoon.
- Lemonade is ready!
Every sip of lemonade tastes great. But wait, where did the sugar go? Did it disappear?
Well, it did not vanish anywhere — it dissolved.

When a solid (like sugar) disappears into a liquid (like water), we say it has dissolved to form a solution. The solid is called the solute, the liquid is the solvent, and the final drink you enjoy is called a solution.
This whole process is termed dissolution, and the ability of a substance to dissolve is called solubility.
What is Solubility?
Solubility can be defined as:
“The maximum amount of a solute that can be dissolved in 100 g of a solvent at a specific temperature to form a saturated solution”.
Here, the solution is in dynamic equilibrium.
Dynamic Equilibrium
Dynamic equilibrium occurs in a saturated solution. It is established when the rate at which solute particles dissolve equals the rate at which they recrystallize (precipitate).
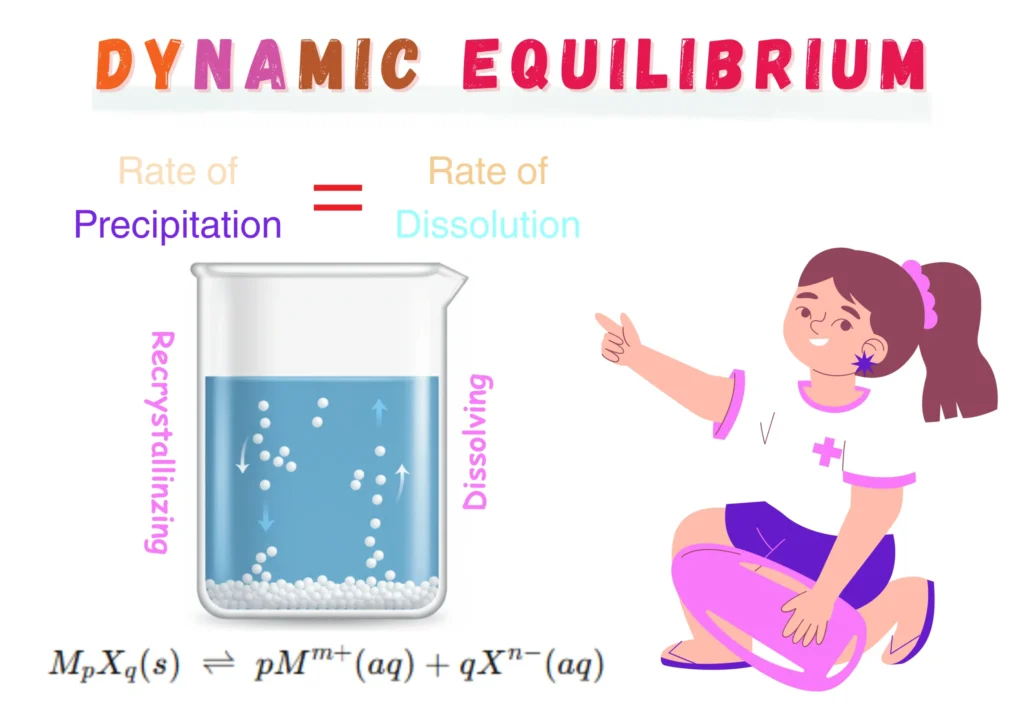
Although particles are continuously dissolving and recrystallizing, the overall concentration of the solute remains constant.
3 Factors Affecting Solubility
The solubility of a substance depends on several factors, including:
1. Effect of Temperature on Solubility
Temperature has a profound effect on solubility. As a general trend, two cases are mentioned here.

Solids in Liquids
In most cases, solubility increases with an increase in temperature.
Solubility ∝ Changes in Temperature
This means that more solute can dissolve in hot water than in cold water.
You can try this at home by mixing sugar in a hot glass of water and a cold glass of water. A hotter glass of water will dissolve more sugar than a colder glass of water.
Examples of Solids and Their Solubility Behaviour
| Compound | Solubility Trend with Temperature |
| Potassium Nitrate (KNO₃) | Increases sharply |
| Silver Nitrate (AgNO₃) | Increases |
| Potassium Chloride (KCl) | Increases |
| Copper Sulphate (CuSO₄) | Increases |
| Sodium Nitrate (NaNO₃) | Increases |
| Sodium Chloride (NaCl) | Barely changes |
| Calcium Hydroxide (Ca(OH)₂) | Decreases |
| Lithium Carbonate (Li₂CO₃), | Decrease |
| Cerium Sulphate (Ce₂(SO₄)₃) | Decrease |
Gases in Liquids
The solubility of gases decreases with increasing temperature. This is why cold soda fizzes more when opened.
Note that warmer temperatures make gases escape faster.
2. Nature of Solute & Solvent
There is a simple idea in chemistry that helps us understand this:
“Like dissolves like”.
This means that substances with similar properties tend to mix well.
Examples
Polar Substances
Water is a polar substance, meaning it has tiny electric charges. So, it can only dissolve other polar or ionic substances (e.g., NaCl, K₂CO₃, CuSO₄, sugar).
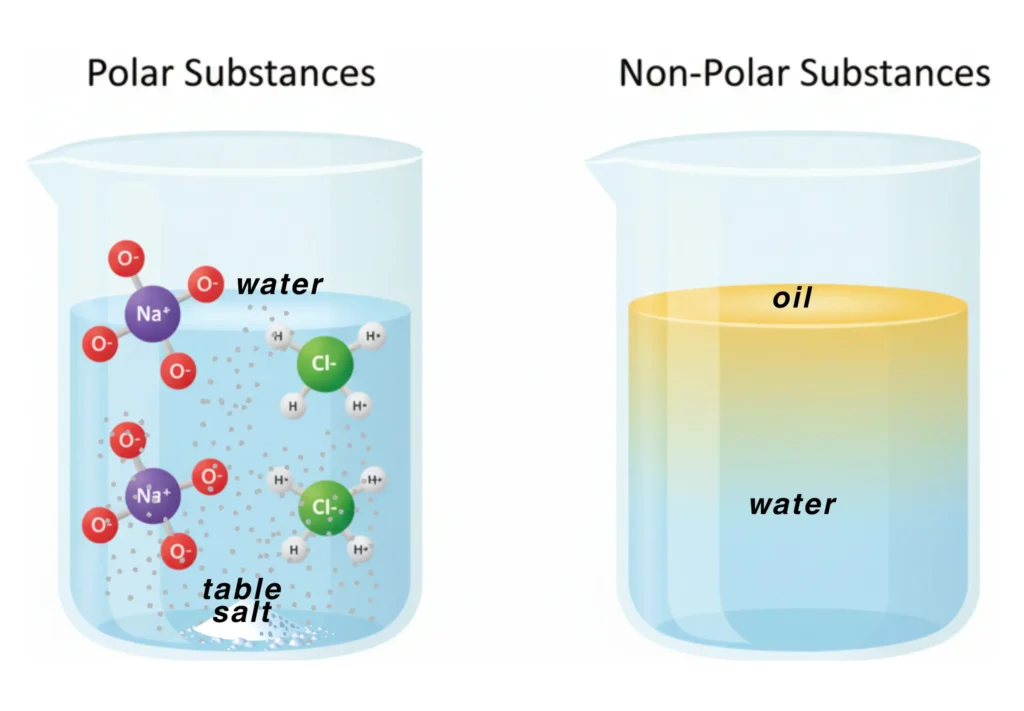
Non-Polar Substances
Oil is a non-polar substance, meaning it does not have tiny charges. So, it will not mix with water. However, being a non-polar substance, it will dissolve easily with other non-polar substances (e.g., wax, grease, ether, naphthalene, or CCl₄).
This is why oil floats on top of water and does not mix.
3. Solute–Solvent Interactions
Dissolving a solute in a solvent requires overcoming the intermolecular forces within both the solute and the solvent. The extent to which a solute dissolves depends on the strength and compatibility of these interactions.
Factors Influencing the Interactions
There are mainly 3 factors that limit the solubility of a solute in a solvent. The factors are:
- Solute–Solute Attractions
- Solvent–Solvent Attractions
- Solute–Solvent Attractions
So, the solubility rule is:
Solute–Solute & Solvent–Solvent Attraction < Solute–Solvent Attraction → Soluble
Solute–Solute & Solvent–Solvent Attraction > Solute–Solvent Attraction → Insoluble
Complete Solubility Requirement
Most solutes are solids, and a major portion of those solids are ionic in nature. These solute ions are held together by strong electrostatic forces in a crystal lattice. For such solids to dissolve:
- Solvent molecules must disrupt the ionic lattice.
- The ions must then be surrounded (hydrated or solvated) by solvent molecules.
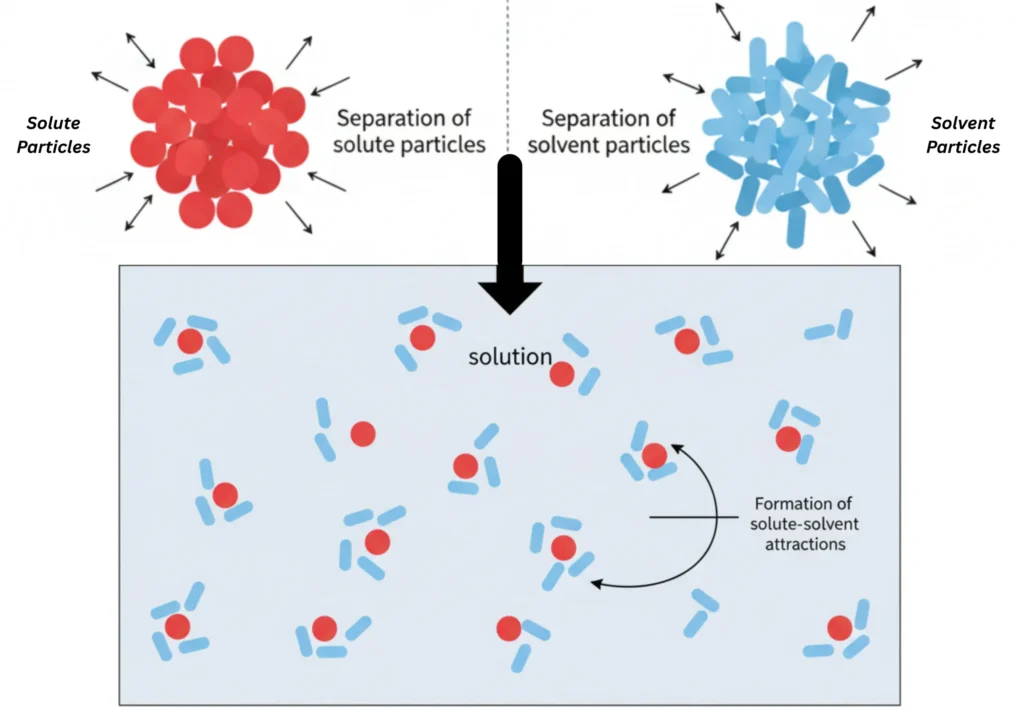
For a solution to be completely soluble, the following 3 steps must occur:
i. Separation of solute particles
ii. Separation of solvent particles
iii. Formation of solute–solvent attractions
Example | Ionic Solids and Dissolution
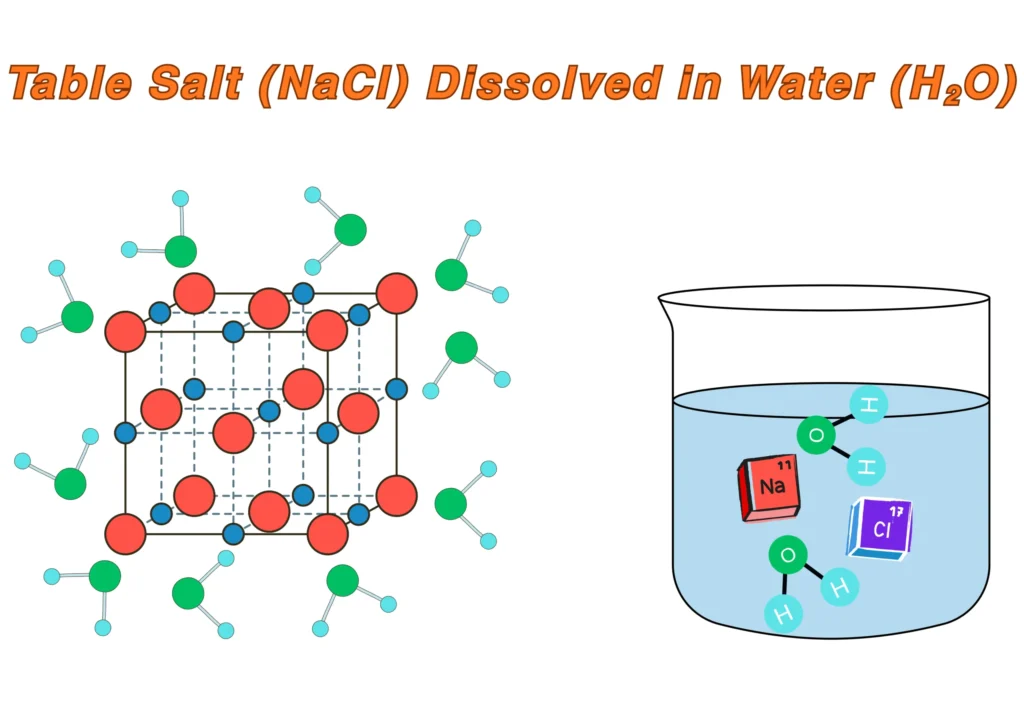
For instance, table salt (NaCl) is dissolved in water (H2O) as follows:
- The polar water molecules interact with Na⁺ and Cl⁻ ions.
- The positive end of water (H) faces Cl⁻, and the negative end (O) faces Na⁺.
- The strong ion–dipole attractions pull the Na⁺ and Cl⁻ ions away from the crystal.
- The ions become surrounded by water molecules and go into solution.
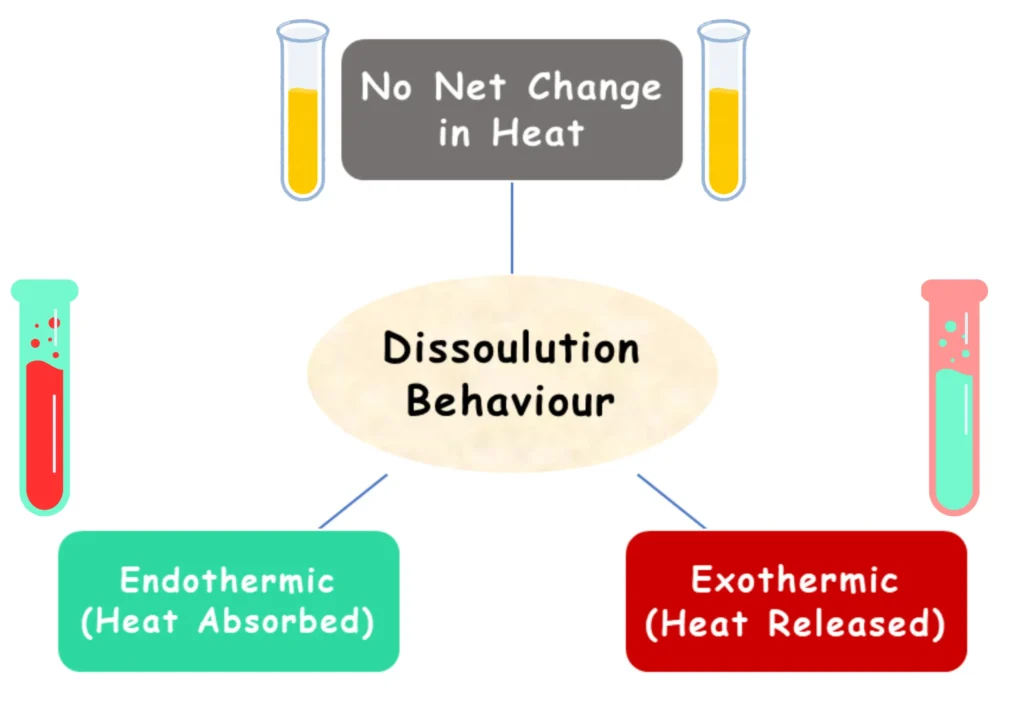
Endothermic Dissolution vs Exothermic Dissolution vs No Net Heat Change | The Science behind Dissolution
When a solute dissolves in a solvent, energy changes take place. There are 3 types of energy behaviour during dissolution:
| Endothermic Dissolution | No Net Heat Change | Exothermic Dissolution |
| The process of dissolving absorbs heat from the surroundings. | No noticeable heat is absorbed or released. | The process releases heat during dissolution. |
| The test tube feels cold during dissolution. | The test tube feels neither warm nor cold. | The test tube becomes warm. |
| Solubility increases with temperature. | Temperature has little effect on solubility. | Solubility decreases with temperature. |
| Example: KNO₃, NaNO₃, KCl | Example: NaCl | Example: Li₂SO₄, Ce₂(SO₄)₃ |
This shows, different compounds respond differently to temperature changes, leading to distinct solubility patterns in water at a given temperature.
Solubility Curves
A solubility curve is a graph of solubility (g solute/100 g solvent) versus temperature (°C). It shows how the solubility of substances varies with temperature.
There are mainly 2 types of solubility curves that are discussed below.
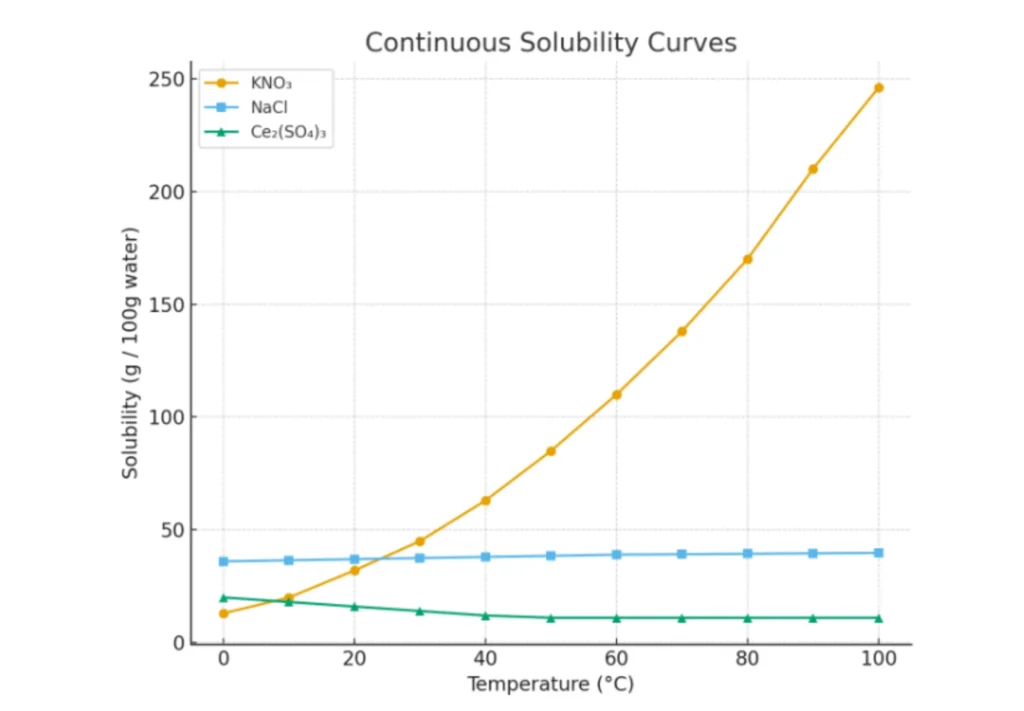
Continuous Solubility Curve
The substances that show a continuous change in solubility are termed a continuous solubility curve. These are smooth and monotonic changes, and the curve appears as a single continuous line.
Example
- KNO₃, KClO₃, and Pb(NO₃)₂ show a continuous increase with temperature.
- NaCl shows only a slight increase.
- Ce₂(SO₄)₃ shows a decrease, which then levels off at higher temperatures.
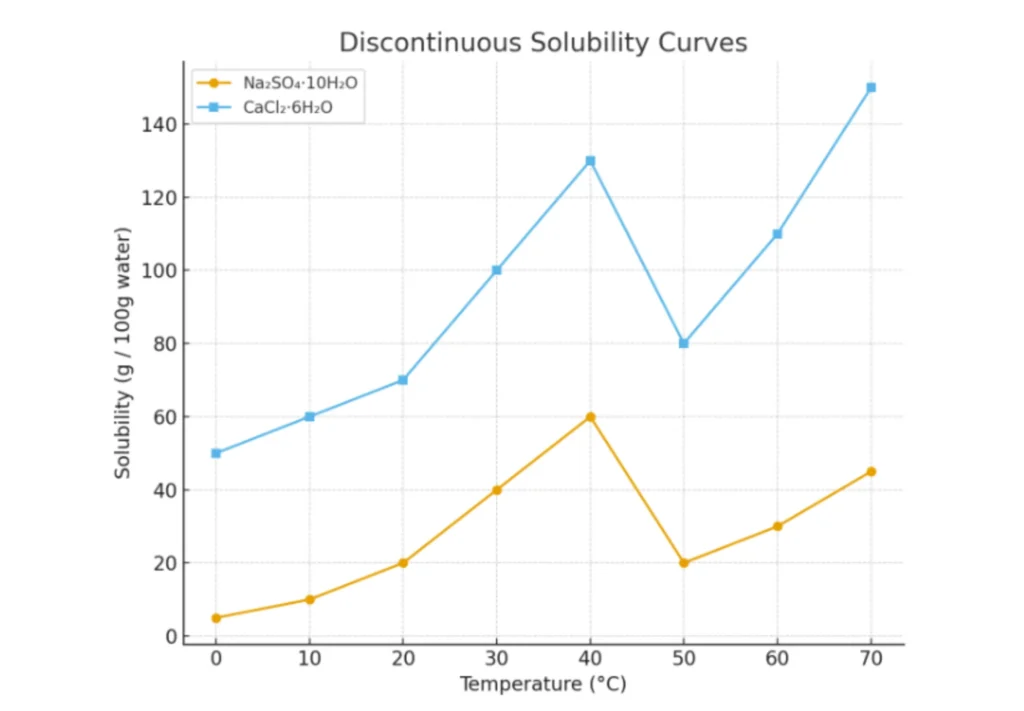
Discontinuous Solubility Curves
The curves that show sudden changes in solubility due to changes in hydration (water of crystallisation) are known as discontinuous solubility curves. These curves appear as a combination of multiple solubility lines.
Example
- Na₂SO₄·10H₂O
- CaCl₂·6H₂O
Conclusion
Here, you saw what solubility is and how it is affected by different factors, especially the effect of temperature on the solubility of solids and gases.
Understanding solubility and heat changes during solute dissolution helps to explain why solubility increases or decreases. Solubility curves and fractional crystallisation are also useful tools for studying and applying solubility behaviour.
Frequently Asked Questions (FAQs)
What is solubility in chemistry?
The maximum amount of solute that dissolves in 100 g of solvent at a specific temperature.
What are 3 factors affecting solubility?
- Temperature
- Nature of solute & solvent
- Solute–solvent interaction
How does NaCl dissolve in water?
Polar molecules of water pull apart Na⁺ and Cl⁻ ions, forming a solution.
Why do different compounds have different solubilities in water at a particular temperature?
Due to differences in bonding, energy changes, and solute–solvent interactions.
Why cannot NaCl be crystallised like KNO₃?
The solubility of NaCl changes very little with temperature, making it hard to recover by cooling. In contrast, the solubility of KNO₃ increases significantly with heat, so cooling a hot, saturated solution leads to visible crystallisation.
What happens if solute–solute forces are stronger?
The solute will not dissolve.
What if solute–solvent forces are stronger?
A solution will form.
Why does iodine dissolve in CCl₄ but not in water?
Because both iodine and CCl₄ are non-polar and water is polar (“like dissolves like”).
Why does KNO₃ make the test tube cold?
Dissolution of KNO3 is an endothermic process (absorbs heat).
What does “like dissolves like” mean?
Polar dissolves polar (e.g., salt in water); non-polar dissolves non-polar (e.g., oil in hexane).
How do intermolecular forces affect solubility?
Solute dissolves only if solute–solvent attraction is stronger than internal forces.

