“Elements and the classification of elements” is a beginner’s guide to the fundamental building blocks of everything around and within us.
Table of Contents
Introduction
Take a deep breath! The air you are inhaling is full of elements—oxygen, nitrogen, even a bit of argon. They are invisible, but they are everything.
However, not all elements are invisible, for instance, an iron sword used a couple of hundred years ago in wars, the gold jewellery females adorn themselves with, or the electrical wire of appliances, etc.
From the blood in your veins to the soil under your feet, elements are the original ingredients that build our bodies, our homes, and the entire planet.
But what exactly are elements? Why do they matter so much? And how did we even discover them?

What Is an Element?
An element can be defined as:
“the simplest form of a pure substance and cannot be broken down into anything simpler by ordinary chemical means.”
Key Terms
In the definition of elements, two terms required special attention:
- Pure Substance
- Ordinary Chemical Means
Pure substance means, the matter is made entirely of one kind of atom, having the same atomic number (which represents the number of protons in the nucleus).
Ordinary chemical means, standard chemical processes or reactions (like burning, rusting, or dissolving) that do not alter the atomic nucleus, unlike nuclear processes.
A Brief History of Elements

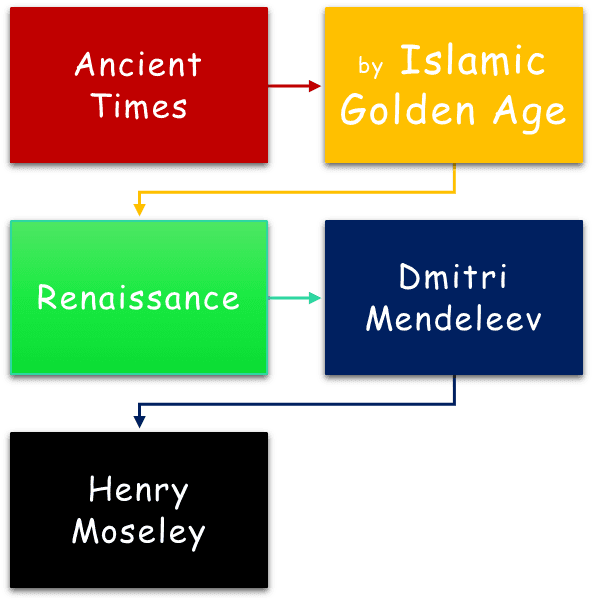
Ancient cultures like the Greeks, Indians, and Chinese saw the world as made of four elements—fire, water, air, and earth. They used this classification to explain nature philosophically.
By the Islamic Golden Age, only nine elements were known. These were classified based on observable traits rather than any atomic model. The reason is the proto-scientific nature of human understanding at the time, which was heavily influenced by the Sulphur-Mercury theory.
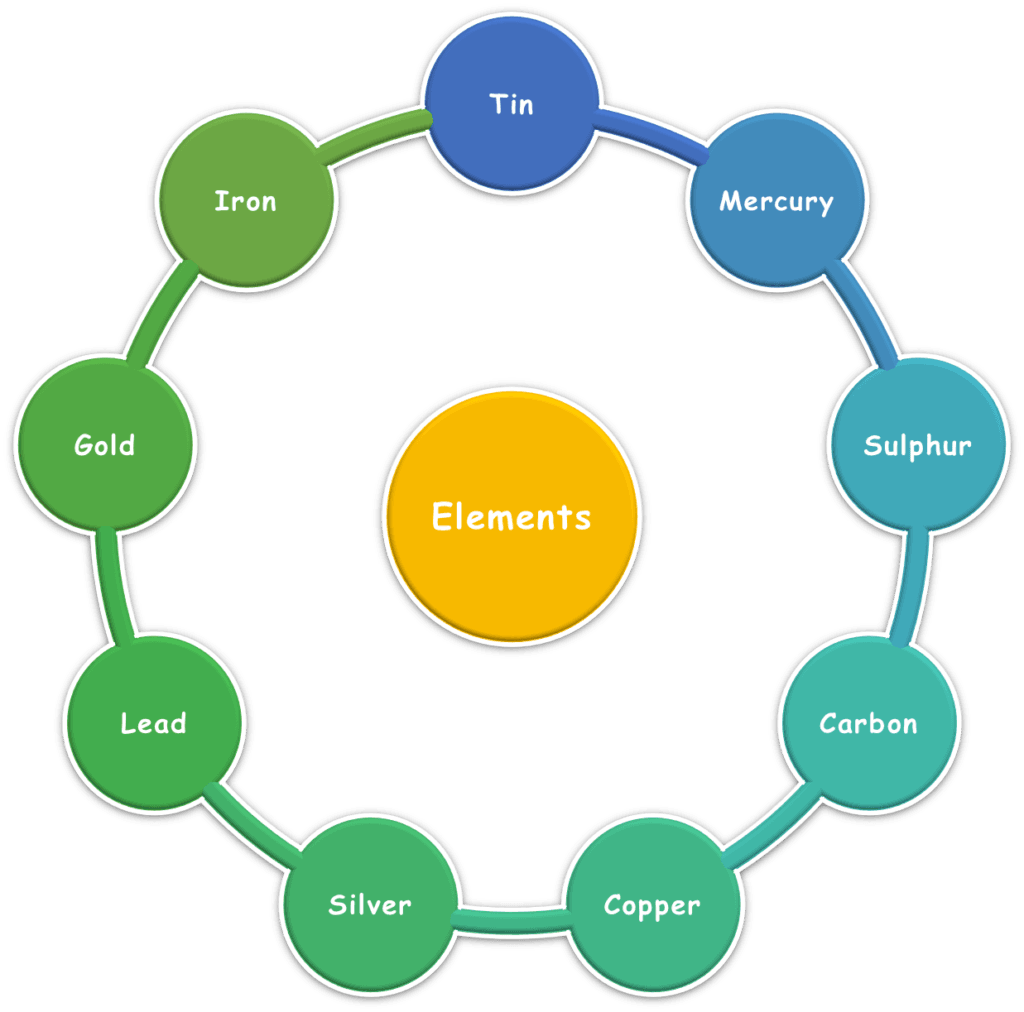
After the Renaissance, Döbereiner’s triads, Newlands’ octaves, and Lothar Meyer’s graphs (atomic mass vs atomic volume) revealed repeating patterns in element properties.
In 1869, Mendeleev arranged elements by atomic mass, predicting missing ones with accuracy. Moseley later corrected the table by atomic number, forming the modern periodic law.
Today, 118 elements are known to the world, of which 92 are natural and the rest are synthetic. They are organised by atomic number, electron configuration, and group behaviour.
The first artificial element created by the scientist in the laboratory was Technetium (Tc). It has also been detected in the spectra of certain stars, indicating that nature can create it under extreme stellar conditions.
This historical context reflects our shift from myth to scientific understanding of the element.
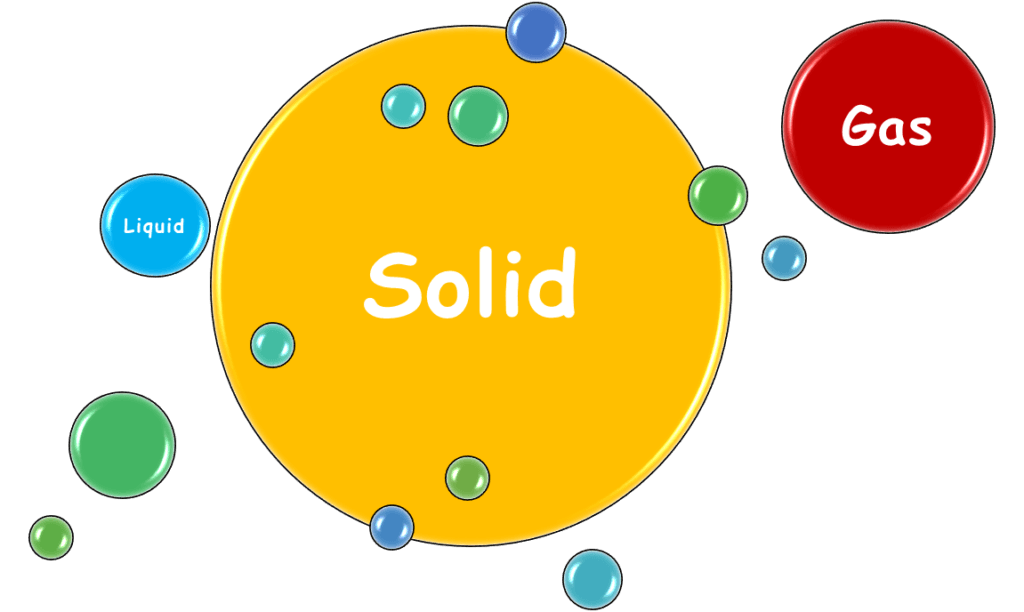
States of Elements
Elements exist in 3 physical states at room temperature: solid, liquid, and gas. These states determine how we use them in various applications. Of the three, solids are the most abundant, while only a handful are gases, and just a few are liquids.
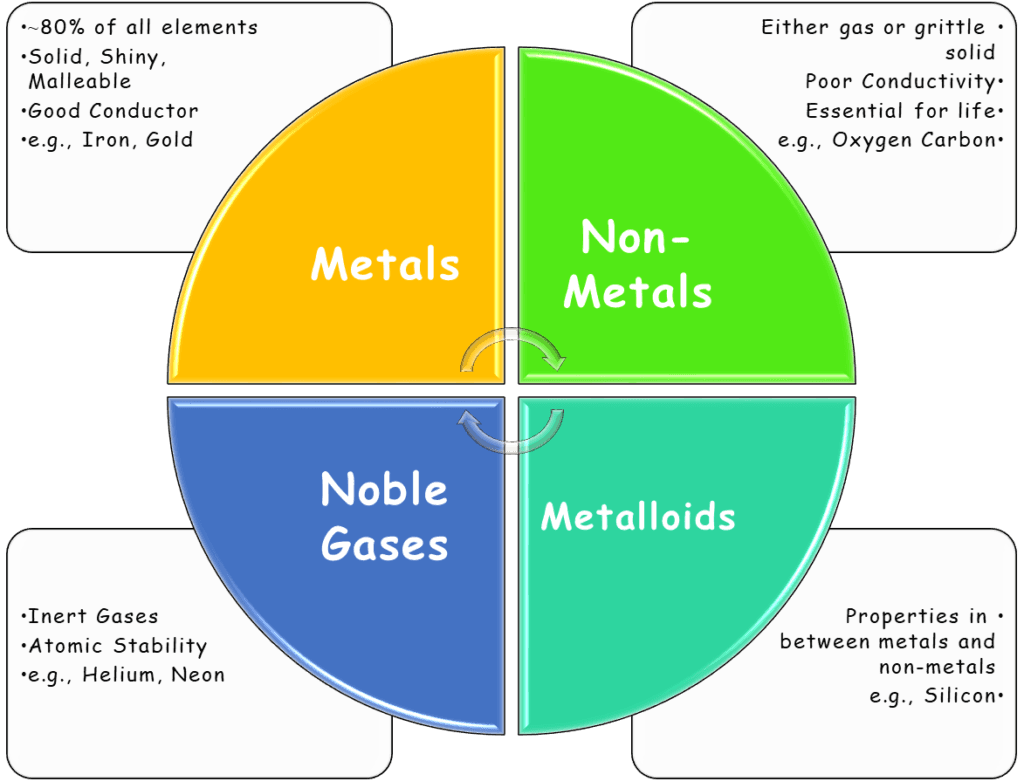
Types of Elements
Based on their properties, elements are grouped into four main categories:
Where Do Elements Exist?
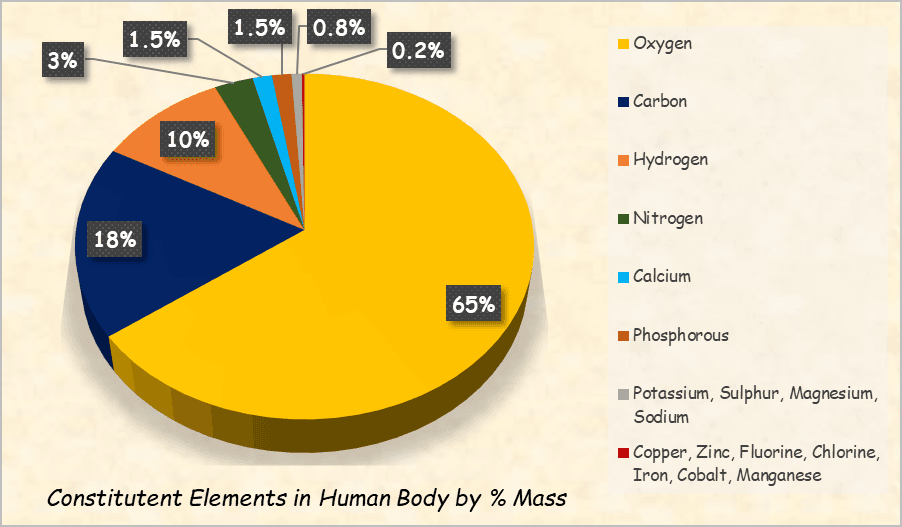
From the heights of the sky to the surface of the Earth and the depths of the oceans, elements are found throughout nature. Some of the major ones are enumerated here:

Why Elements Matter
The following facts prove, “Why do elements matter?”
- Elements power your electronic devices, build your cities, and keep you alive.
- The blood in the veins and the air you breathe are composed of elements.
- Your thoughts rely on sodium and potassium ions.
- Your bones contain calcium and phosphorus.
- Your blood carries iron and oxygen.
Symbols of Elements
Each element is represented by a symbol, which is its abbreviated name. These symbols often derive from Latin, Greek, German, or English names. The symbol of an element contains:
- One capitalised letter, for instance, H (Hydrogen), C (Carbon)
- Two-letter words, where the first is capitalised and the second small, for instance, Ca (Calcium), Na (Sodium — from its Latin name Natrium)
Valency of an Element
The valency of an element is its capacity to combine with another element. It is another key trait of an element. It depends on the number of electrons in the outermost shell and determines how an element will bond with others. For instance:
- Calcium, +2
- Aluminium, +3
- Oxygen, –2
- Chlorine, –1
Conclusion
Once you start recognizing the elements in your life, everything in the universe begins to align. Consider this:
- The spoon in your hand is likely stainless steel—an alloy of iron, chromium, and nickel.
- Your phone screen is made of silicon and rare earth elements.
- Even your breath is sustained by oxygen and nitrogen.
This understanding is not just academic—it is part of our shared human heritage. It laid the foundation for modern chemistry and connects us to the long journey of scientific discovery and human resilience.
So the next time someone asks what you are made of, you can clearly and proudly say:
“I am a living, breathing combination of elements.”
Frequently Asked Questions (FAQs)
Name the elements represented by the following symbols: Hg, Au, Fe, Ni, Co, W, Sn, Na, Ba, Br, Bi.
- Hg – Mercury
- Au – Gold
- Fe – Iron
- Ni – Nickel
- Co – Cobalt
- W – Tungsten
- Sn – Tin
- Na – Sodium
- Ba – Barium
- Br – Bromine
- Bi – Bismuth
Name a solid, a liquid, and a gaseous element that exists at room temperature.
- Solid – Iron (Fe)
- Liquid – Mercury (Hg)
- Gas – Oxygen (O₂)
Which is the most abundant element occurring in the oceans?
Oxygen is the most abundant element (86%) in the oceans, primarily as part of water (H₂O) and dissolved oxygen.
Which one of the elements is found in the most abundance in the Earth’s crust?
Oxygen is the most abundant element in the Earth’s crust (about 47 % by weight), mainly in oxides and silicates.
Which is the third abundant gas found in the Earth’s atmosphere?
Argon (Ar) is the third most abundant gas, after nitrogen and oxygen.
Explain why hydrogen and oxygen are considered elements.
Hydrogen and oxygen are considered elements because they consist of only one type of atom, and cannot be broken down into simpler substances by ordinary chemical means. Each has a unique atomic number: hydrogen (1), oxygen (8).
What is the significance of the symbol of an element?
The symbol of an element is a standardised, short representation (usually 1–2 letters) used in chemical equations and formulas.
- It simplifies writing and communication in chemistry.
- For instance, H for hydrogen, O for oxygen, Na for sodium.
What do you know about elements and the classification of elements? Or define an element and classify the elements with examples.
An element is a pure substance that cannot be broken down into simpler substances by ordinary chemical means. It consists of only one type of atom.
Classification of Elements:
1. Metals
- Good conductors, malleable, shiny
- Examples: Iron (Fe), Copper (Cu), Aluminium (Al)
2. Non-Metals
- Poor conductors, brittle, often gas
- Examples: Oxygen (O), Nitrogen (N), Sulphur (S)
3. Metalloids (Semi-metals)
- Have properties of both metals and non-metals
- Examples: Silicon (Si), Arsenic (As)
4. Noble Gases
- Inert, colourless gases
- Examples: Helium (He), Neon (Ne), Argon (Ar)
