The differences between Ultraviolet Radiations vs X Rays vs Gamma Rays, their wavelength, frequency ranges, properties, applications, and real-world uses are discussed here.
Table of Contents
Introduction
When we look at the grand span of the electromagnetic spectrum (EM), we find an endless sequence of waves. These waves include radio waves that stretch thousands of kilometres to gamma rays that are fiercely energetic and vibrate trillions of times per second.
In previous discussions, we explored the infra-region (the gentler, low-energy side) containing:
- Infrared
- Microwaves
- Radio Waves

Now, we move to the other extreme — the ultra-region. The most prominent things about this region are:
- Energy rises sharply
- Wavelengths shrink to atomic scales
- Photons carry enough punch to alter molecules
- Penetrate matter, and reveal the structure of the universe itself
This “ultra-region” is a learning construct that lies beyond the violet edge of visible light. It is a conceptual division used here for clarity, not a standard scientific term. It covers:
- Ultraviolet (UV)
- X-Rays
- Gamma Rays (γ)
These waves are the tools in the transforming contemporary world that can create, destroy, heal, and illuminate the unseen.

Core Physical Principles behind the Ultra-Region
The whole electromagnetic spectrum deals with 3 core principles, including:
1. Wave–Speed Relation (![]() )
)
2. Energy of a Photon (![]() )
)
3. A Continuous Spectrum without Walls
These features help to understand the key physical ideas that explain why these waves behave the way they do.
What Sets Them Apart? | Infra–Region vs Ultra–Region
Both regions follow the same laws of light, but they differ in how those laws appear.
- In the infra-region, light behaves mostly as a continuous wave, with effects explained by classical physics.
- In the ultra-region, each photon carries enough energy to alter electronic states, eject electrons, or even affect atomic nuclei.
The ultra-region thus marks the threshold where light and matter reveal their deepest quantum behaviour. In a nutshell, the quantum nature of light dominates.
The Ultra-Region of the EM Spectrum
The ultra-region occupies the high-frequency, high-energy end of the spectrum. It extends from the near-ultraviolet edge of visible light to gamma radiation.
| Property | Approximate Range |
| Wavelength | ~400 nm → <0.01 nm |
| Frequency | ~7.5×10¹⁴ Hz → >10²⁰ Hz |
| Photon Energy | ~3 eV → >1 MeV |
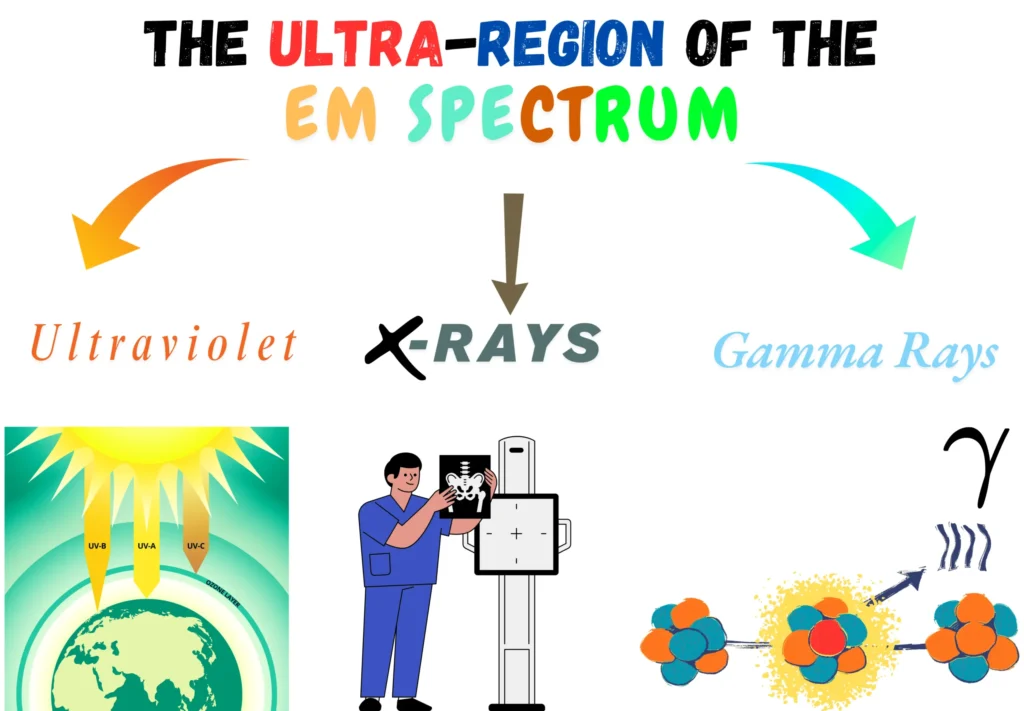
Here, you will explore 3 distinct types of electromagnetic radiation within the ultra-region. Each type is defined by its unique origin, interaction, and effect on matter.
1. Ultraviolet (UV) Radiation
Range: ~400 nm – 10 nm
Frequency: ~7.5×10¹⁴ – 3×10¹⁶ Hz

Characteristics
Ultraviolet lies immediately above the violet limit of visible light. Although invisible to the human eye, UV has a profound influence on living systems and materials. It causes fluorescence, chemical reactions, and biological changes.
Division of Ultraviolet Radiation
The UV radiation can be categorised into the following:
- UVA (Near UV)
- UVB (Middle UV)
- UVC (Far UV)
- VUV (Vacuum UV – sometimes grouped under UVC)
A comparison of these radiations is given here:
| Property | UVA | UVB | UVC | VUV |
| Wavelength range (nm) | 400–315 | 315–280 | 280–100 | 200–10 |
| Photon energy (eV) | 3.1–3.9 | 3.9–4.4 | 4.4–12.4 | 6.2–124 |
| Reaches Earth’s surface? | Yes | Partially | No | No (absorbed by air) |
| Atmospheric absorption | Weak (passes through) | Partial (ozone absorbs some) | Strong (blocked by ozone) | Complete (absorbed by all gases) |
| Primary interactions | Excites molecules | Breaks some bonds, damages DNA | Ionizes molecules, breaks bonds | Strong ionization and dissociation |
| Typical biological effects | Tanning, skin aging | Sunburn, DNA mutation, vitamin D production | Germicidal, highly damaging to tissue | Lethal to cells, not naturally encountered |
| Common uses | Black lights, UV curing, fluorescence | Medical phototherapy, sterilisation | Germicidal lamps, ozone generation | Spectroscopy, semiconductor cleaning |
| Nature of radiation | Non-ionising | Weakly ionising potential | Ionising | Strongly ionising |
Applications
- Sterilisation & Disinfection (UV-C lamps in hospitals and water systems)
- Tanning Devices & Vitamin D Production
- Fluorescence & Forensic Analysis
- Semiconductor Photolithography
Hazards / Protection
- Overexposure can cause skin burns, cataracts, and DNA damage.
- Protection involves sunscreen, UV-blocking eyewear, and ozone layer conservation.
2. X-Rays
Range: ~10 nm – 0.01 nm
Frequency: ~3×10¹⁶ – 3×10¹⁹ Hz

Characteristics
X-rays occupy the middle of the ultra-region. Their discovery by Wilhelm Roentgen (1895) opened an entirely new window into the hidden world.
They are usually classified as:
- Soft X-rays (lower energy, absorbed by tissues)
- Hard X-rays (higher energy, penetrate deeply)
Generation of X-Rays
X-rays are produced when high-energy electrons interact with matter. This most commonly takes place within an X-ray tube.
Inside the tube, electrons are accelerated through a high voltage and directed onto a metallic target (typically tungsten). When these fast-moving electrons collide with the target atoms, they decelerate, and their kinetic energy is converted into electromagnetic radiation.
This conversion into electromagnetic radiation occurs through two primary mechanisms:
- Bremsstrahlung
- Characteristic Radiation
| Property | Bremsstrahlung Radiation | Characteristic Radiation |
| Origin | Produced when high-speed electrons are decelerated or deflected by the electric fields of atomic nuclei. | Produced when an incoming electron ejects an inner-shell electron, and an outer electron transitions down to fill the vacancy. |
| Type of Spectrum | Continuous — The energy of emitted photons varies continuously depending on electron–nucleus interactions. | Discrete — Specific, well-defined energy lines corresponding to atomic shell transitions. |
| Energy Dependence | Depends mainly on the accelerating voltage of the X-ray tube. | Depends on the atomic energy levels of the target element. |
| Element Specificity | Not element-specific — Occurs for any target material | Element-specific — Each element emits a unique set of characteristic lines. |
| Typical Role | Forms the broad background or base of the X-ray spectrum. | Appears as sharp peaks superimposed on the continuous spectrum. |
| Example Application | Used in general radiography and imaging, where a continuous spectrum is required. | Used in X-ray fluorescence (XRF) and elemental analysis. |
Applications
- Security Scanners & Industrial Inspection
- Medical Imaging (CT scans, dental X-rays)
- X-ray Crystallography (molecular and crystal structure analysis)
Hazards / Shielding
- Prolonged or intense exposure can damage tissues and increase cancer risk.
- Protection uses lead aprons, walls, or glass to absorb stray radiation.
3. Gamma Rays (γ-Rays)
Range: <0.01 nm
Frequency: >3×10¹⁹ Hz
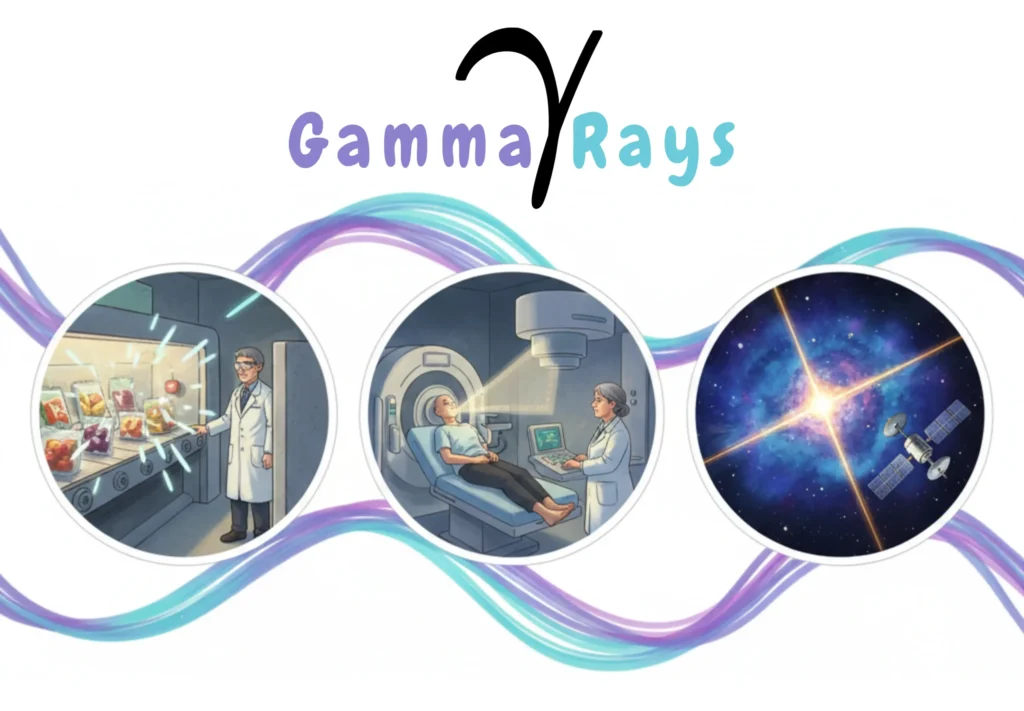
Characteristics
Gamma rays are the most energetic and penetrating form of electromagnetic radiation. They emerge from nuclear transitions, radioactive decay, and cosmic phenomena.
Sources of γ-Rays
- Radioactive Decay (Cobalt-60, Cesium-137)
- Cosmic Events (supernovae, pulsars, black holes)
- Nuclear Reactions and Matter–Antimatter Events
Applications
- Food Irradiation
- Cancer Radiotherapy
- Sterilization of Medical Instruments
- Gamma Astronomy & Space Science
Hazards / Protection
- Gamma rays can penetrate deeply into living tissue.
- Protection requires dense materials (lead, tungsten) or thick concrete walls and precise dose regulation.
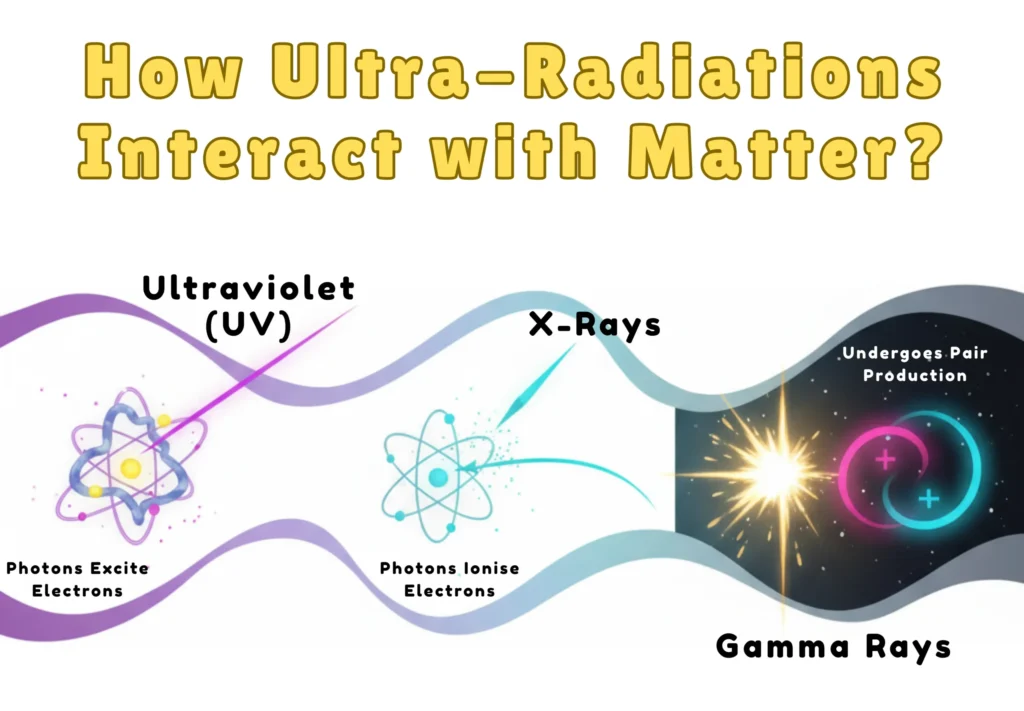
How Ultra-Radiations Interact with Matter?
Across the ultra-region, from UV to X-rays to gamma rays, the way light interacts with matter shifts dramatically. It all depends on the energy of the photon.
- Ultraviolet (UV): Photons excite electrons within atoms and molecules, triggering electronic transitions and photochemical reactions such as ozone formation and DNA mutation.
- X-Rays: Higher-energy photons can ionise atoms through the photoelectric effect or Compton scattering, and at extreme energies, even cause pair production.
- Gamma Rays: These are the most energetic photons. They primarily undergo pair production, where energy transforms into an electron–positron pair, a striking display of E = mc².
In short:
- UV excites electrons, X-rays eject them, and gamma rays create them.
- These interactions reveal the quantum, particle-like nature of high-frequency light.
Differences between Ultraviolet Radiations vs X Rays vs Gamma Rays
| Property | Ultraviolet (UV) | X-Rays | Gamma Rays (γ) |
| Wavelength Range | 400 – 10 nm | 10 – 0.01 nm | < 0.01 nm |
| Frequency Range | 7.5×10¹⁴ – 3×10¹⁶ Hz | 3×10¹⁶ – 3×10¹⁹ Hz | >3×10¹⁹ Hz |
| Photon Energy | 3 – 100 eV | 100 eV – 100 keV | >100 keV |
| Penetration Ability | Low–Moderate | Moderate–High | Very High |
| Typical Uses | Sterilisation, fluorescence, tanning | Medical imaging, crystallography | Radiotherapy, astrophysics |
| Protection | Sunscreen, ozone layer | Lead aprons, barriers | Lead/concrete shielding |
Ethical Considerations
Science and medicine must balance discovery with responsibility. The same radiation that saves lives can also cause harm if misused. Ethical use requires strict control, monitoring, and respect for biological limits.

Conclusion
The ultra-region marks the quantum frontier of the electromagnetic spectrum. Here, the light no longer behaves purely as a wave; it begins to act as a corpuscle of energy.
From ultraviolet sparks of life to the gamma fire of cosmic events, the ultra-region of the EM spectrum shows us that energy is the architect of reality. If they are harnessed wisely, they can heal, reveal, and redefine our understanding of the unseen universe.
Frequently Asked Questions (FAQs)
What is the ultra-region of the electromagnetic spectrum?
The ultra-region refers to the high-energy end of the electromagnetic spectrum, which includes ultraviolet (UV) rays, X-rays, and gamma rays. These radiations have short wavelengths, high frequencies, and photons with enough energy to ionize matter.
How do ultraviolet rays, X-rays, and gamma rays differ?
Ultraviolet rays have the longest wavelengths and lowest energy, X-rays have higher energy and shorter wavelengths, while gamma rays are the most energetic and penetrate the deepest into matter.
What is the wavelength range of ultraviolet, X-rays, and gamma rays?
- Ultraviolet (UV): 400–10 nm
- X-rays: 10–0.01 nm
- Gamma rays: <0.01 nm
Which radiation is more harmful — UV, X-ray, or gamma ray?
Gamma rays are the most harmful due to their extremely high energy and penetrating power. X-rays are moderately harmful, while most UV rays are less dangerous but can still cause skin damage and DNA mutations.
What are the main sources of UV rays, X-rays, and gamma rays?
- UV Rays: The Sun and artificial UV lamps.
- X-Rays: X-ray tubes and particle accelerators.
- Gamma Rays: Radioactive decay, nuclear reactions, and cosmic phenomena.
How do UV rays, X-rays, and gamma rays interact with matter?
- UV rays excite or ionise atoms.
- X-rays eject electrons via the photoelectric and Compton effects.
- Gamma rays can create matter through pair production and convert energy into an electron–positron pair.
What are the common uses of ultraviolet, X-rays, and gamma rays?
- UV: Sterilisation, tanning, fluorescence.
- X-Rays: Medical imaging, crystallography, security scanning.
- Gamma Rays: Cancer radiotherapy, sterilisation, and astrophysics research.
Why are X-rays and gamma rays considered ionising radiation?
Both X-rays and gamma rays have photon energies high enough to knock electrons out of atoms, creating ions. This ionizing ability is what makes them powerful for imaging and dangerous in high doses.
What protective measures are used against these radiations?
- UV: Sunscreen and UV-blocking lenses.
- X-rays: Lead aprons or barriers.
- Gamma Rays: Thick concrete or dense metal shielding, such as lead or tungsten.
Why are ultraviolet, X-rays, and gamma rays important in science and technology?
These radiations help us explore both microscopic and cosmic worlds — from detecting molecular structures and diagnosing diseases to studying nuclear reactions and distant stars.

