To understand what is a solution in chemistry and the 3 classifications of solutions is crucial for beginners to delve deeper into the study of homogeneous mixtures.
Table of Contents
Introduction
From the air we breathe to the drinks we sip, solutions are an integral part of both our daily lives and the science of chemistry. But what exactly is a solution? How do they work, and why are they so important?
What is a Solution?
A solution can be defined as:
“A homogeneous mixture of two or more substances that exists in a single phase.”
Here are 3 things to be noticed:
- Solutions are homogeneous mixtures, meaning they look the same throughout.
- The number of substances would be more than one and cannot be distinguished from one another with the naked eye.
- Single phase implies, there are no visible boundaries and the entire mixture exists as a single entity (solid, liquid, or gas).
Examples
- Sugar water: A classic liquid solution where sugar dissolves completely in water.
- Air: A gaseous solution of nitrogen, oxygen, carbon dioxide, and more.
- Brass: A solid solution where zinc is mixed with copper.
Solution vs Pure Substance
One easy way to distinguish a solution from a pure substance is through evaporation:
- If the liquid leaves no residue, it is pure.
- If something is left behind, it was a solution.
Components of a Solution
Every solution has two basic parts:
- Solute: The component in smaller quantity, which gets dissolved.
- Solvent: The component present in the largest quantity and dissolves the solute.
Example
In a sugar solution:
- Solute: Sugar
- Solvent: Water
Binary Solution
When there is just one solute and one solvent, it is called a binary solution.
However, many everyday mixtures—like soft drinks—contain multiple solutes (sugar, carbon dioxide, flavourings, salts) all dissolved in water.
Aqueous Solutions
An aqueous solution is one where water is the solvent. These are the most common in nature and laboratories.
Water as a Universal Solvent
Water is often called the universal solvent because it can dissolve a vast range of substances. For instance, it can dissolve:
- Acids, Bases, and Salts
- Solids, Liquids, and Gases
- Organic and Inorganic Substances
Examples
- Salt water
- Soft drinks
- Sugar water
Sea water is a resource of 92 naturally occurring elements.
The 3 Classifications of Solutions
1. Types of Solutions based on Physical State of Solute and Solvent
Depending on the physical states of solute and solvent— solid, liquid, or gas —, solutions can be classified into 9 types:
| Solute | Solvent | Example |
| Gas | Gas | Air (21% oxygen in 78% nitrogen) |
| Gas | Liquid | Carbon dioxide in water (soda) |
| Gas | Solid | Hydrogen in palladium |
| Liquid | Gas | Water vapour in air (humidity) |
| Liquid | Liquid | Alcohol in water |
| Liquid | Solid | Mercury in silver (dental amalgam) |
| Solid | Liquid | Sugar in water |
| Solid | Gas | Dust particles in the air (aerosols) |
| Solid | Solid | Alloys like brass and bronze |
2. Solutions Based on Saturation
Saturation means how much solute a solution can hold. It depends on temperature and the nature of the solute and solvent. Based on this, solutions are categorised into 3 types:
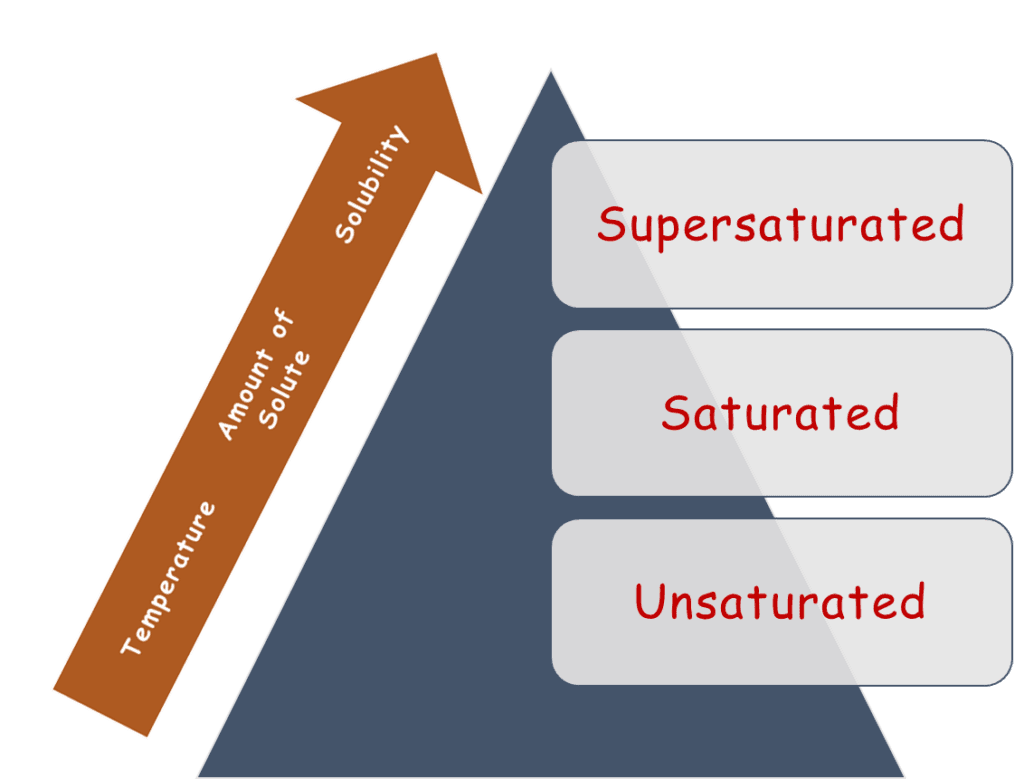
i. Unsaturated Solution
A solution containing less solute than the maximum amount it can hold.
![]()
This solution can still dissolve more solute.
Example
Dissolving 5g of sugar in 100g of water is unsaturated if all the sugar dissolves.
ii. Saturated Solution
A solution containing the maximum amount of solute that can dissolve at a particular temperature.
![]()
In a saturated solution, extra solute remains undissolved.
Example
At 20 °C, 100g of water can dissolve about 36g of salt (NaCl). Anything more settles at the bottom.
Dynamic Equilibrium
At the molecular level, a saturated solution is in dynamic equilibrium. It means the rate at which solute dissolves equals the rate at which it crystallises.
![]()
iii. Supersaturated Solution
A solution containing more solute than is normally possible at an elevated temperature.
It is unstable and can crystallise easily if disturbed.
How Supersaturated Solution Made?
- Dissolve excess solute at a high temperature.
- Cool it slowly without shaking.
Example
Sodium thiosulfate (Na₂S₂O₃) can form supersaturated solutions this way.
Solubility and What Affects It
Saturation of a solution is expressed in terms of its solubility. Different substances have different solubilities. For instance, at 20 °C:
- Solubility of Sugar ~ 203.9g per 100g of water
- Solubility of Salt (NaCl) ~ 36g per 100g of water
This difference arises from the size and nature of the molecules. For example, sugar molecules are larger and require more water molecules to surround and dissolve them.
3. Solutions based on Concentration
Concentration refers to how much solute is dissolved in a given quantity of solvent or solution. Based on the criteria, 2 types of solutions are there: Dilute Solution vs Concentrated Solution.

| Dilute Solution | Concentrated Solution |
| It is a solution that contains only a small amount of solute in a given amount of solvent. | It is a solution that contains a large amount of solute in a given amount of solvent. |
| Tap water is a dilute solution, used in our daily lives. | Brine is a concentrated salt solution used in food preservation. |
How to Adjust the Concentration of Solution?
To adjust the concentration of a solution, either of the following is used:
- The addition of more solute increases concentration.
- The addition of more solvent (dilution) decreases concentration.
Exam Style Questions
Conclusion
From the atmosphere around us to the beverages we enjoy and the metals in our gadgets, solutions are everywhere. They are important not just to chemistry but to daily life.
Understanding:
- What makes a solution?
- How do solutes and solvents interact?
- How do saturation and solubility work?
- Differences between dilute and concentrated solutions.
…is the key to unlocking deeper concepts in chemistry and beyond. Whether you are mixing a sports drink or formulating industrial alloys, you are working with solutions.
Frequently Asked Questions (FAQs)
What is a solution in chemistry? Write 3 classifications of solutions?
A solution is a homogeneous mixture of two or more substances where the composition is uniform throughout.
The 3 classifications of solutions are:
- Based on physical state: Solid, liquid, and gaseous solutions.
- Based on saturation: Unsaturated, saturated, and supersaturated solutions.
- Based on concentration: Dilute and concentrated solutions.
Write the difference between unsaturated vs saturated vs supersaturated solutions.
- Unsaturated solution: Contains less solute than it can dissolve at a given temperature.
- Saturated solution: Contains the maximum amount of solute that can dissolve at that temperature; extra solute remains undissolved.
- Supersaturated solution: Contains more solute than normally possible at that temperature; unstable and prone to crystallization.
Write the difference between dilute vs concentrated solutions.
- Dilute solution: Has a small amount of solute in a large amount of solvent.
- Concentrated solution: Has a large amount of solute in a small amount of solvent.
How are solutions formed: macroscopically and microscopically?
- Macroscopically, the solute dissolves and disappears, forming a uniform mixture.
- Microscopically, solute particles break down and are surrounded by solvent molecules, forming a stable and uniform system.
Define and explain the following with one example in each case: (a) A homogeneous phase, (b) A concentrated solution, (c) A solution of a solid in a solid
| Term | Explanation |
| A Homogeneous Phase | A phase with uniform composition throughout and no visible boundary. Example: Salt dissolved in water. |
| A Concentrated Solution | A solution containing a large amount of solute. Example: Syrup (sugar in water). |
| A Solution of a Solid in A Solid | A solid solute is uniformly mixed in a solid solvent. Example: Brass (zinc in copper). |
Distinguish between the following pairs as compound or solution: (a) water and salt solution, (b) vinegar and benzene, (c) carbonated drinks and acetone.
| Pair | Compound vs Solution |
| (a) water and salt solution | Water is a compound; salt solution is a solution. |
| (b) vinegar and benzene | Vinegar is a solution (acetic acid in water); benzene is a compound. |
| (c) carbonated drinks and acetone | Carbonated drinks are solutions (CO₂ in water); acetone is a compound. |
Mist is an example of a solution: (a) liquid in gas, (b) gas in liquid, (c) solid in gas, (d) gas in solid
(a) liquid in gas
Which one of the following is a ‘liquid in solid’ solution? (a) sugar in water (b) butter (c) opal (d) fog
(b) butter
When a saturated solution is diluted, it turns into: (a) a supersaturated solution, (b) an unsaturated solution, (c) a concentrated solution, (d) none of these
(b) unsaturated solution
An aqueous solution of ethanol in water may have vapour pressure: (a) equal to that of water, (b) equal to that of ethanol, (c) more than that of water, (d) less than that of water
(c) more than that of water

